Water Absorption in Custom Tableware: A Deep Technical Interpretation for Ceramic Buyers and Manufacturers
1. Deep Definition: What Water Absorption Really Means in Tableware
Water absorption is the percentage of water a fired ceramic body can absorb through its connected pore network after drying and saturation. In laboratory terms, it is a mass-gain measurement; in industrial terms, it is one of the clearest indicators of whether a body is matured, sufficiently vitrified, and structurally reliable for long-term tableware use. ASTM explicitly treats water absorption together with bulk density, apparent porosity, and apparent specific gravity as tools for assessing the degree of maturation of a ceramic body. Source
For custom dinnerware, this metric matters far beyond a lab report. A low absorption body is generally denser, less permeable, less prone to moisture-driven expansion, and better able to support the glaze during dishwasher cycles, thermal shock, and repeated utensil impact. By contrast, a high absorption body often signals underfiring, incomplete vitrification, or unstable body formulation, all of which can shorten service life in hospitality, retail, or OEM projects. ISO 5644, the latest porcelain tableware specification, states that porcelain tableware shall have water absorption below 0.5% when tested by the referenced method. Source
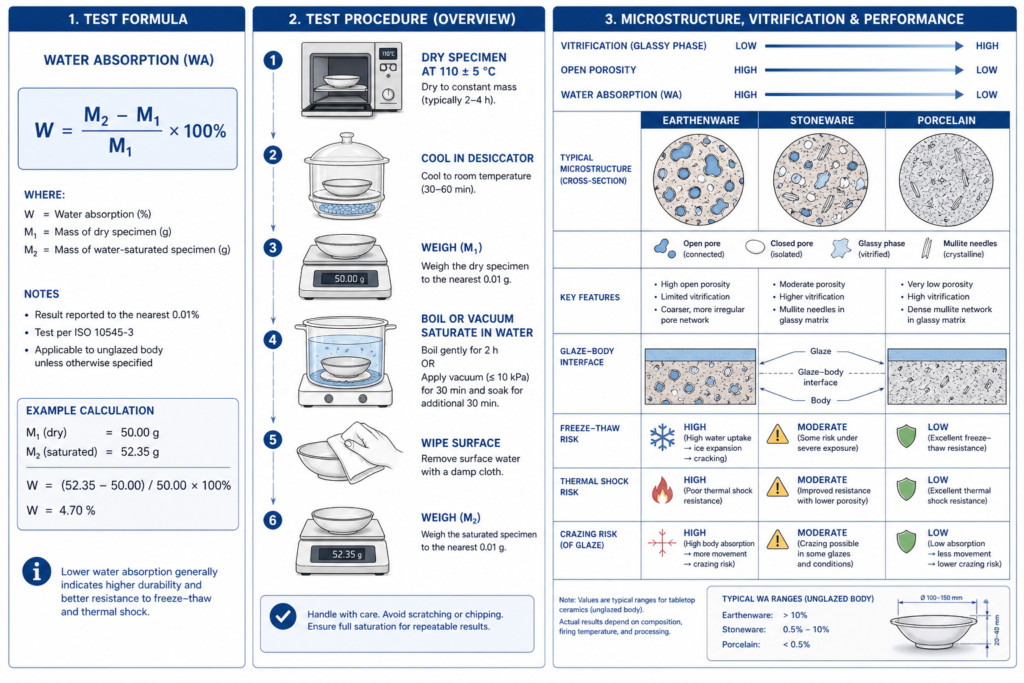
2. The Core Test Formula and What It Physically Represents
The classic expression for water absorption is:
W = ((M2 − M1) / M1) × 100%
Where:
- M1 = dry mass of the specimen after oven drying
- M2 = saturated mass after water immersion / boiling saturation
This equation measures the ratio between absorbed water and the specimen’s dry mass. In other words, it quantifies how much open, connected pore volume remains in the fired body after sintering. The ASTM expression is equivalent in form:
A = ((M − D) / D) × 100
where D is dry mass and M is saturated mass. Source
If a factory wants deeper microstructural insight, water absorption should be read together with apparent porosity and bulk density, which ASTM C373 also provides:
Pa = ((M − D) / (M − S)) × 100
ρb = D / (M − S)
where S is the suspended mass measured while the specimen is immersed in water. This is critical because two bodies may show similar nominal absorption values but differ in pore morphology, density, and crack sensitivity. Source
3. Hard-Core Principle: Why Water Absorption Governs Durability
3.1 Water absorption is a proxy for open porosity
In fired ceramics, water does not enter closed pores; it enters only the connected capillary network. Therefore, absorption is not merely a “wetting” issue. It is a structural indicator of how much of the ceramic body remains open to fluid ingress after firing. The lower the open porosity, the fewer pathways exist for moisture penetration, staining, detergent attack, and delayed moisture expansion. ASTM explicitly links porosity and density measurement to the degree of ceramic maturation. Source
From a durability standpoint, this matters because open porosity acts as a flaw network. Pores reduce effective load-bearing area, concentrate local stress, and weaken the body-glaze system under mechanical and thermal cycling. In tableware, that translates into greater risk of edge chipping, delayed crazing, stain pickup, odor retention in unglazed areas, and premature failure in repeated dishwasher or microwave-related use.
3.2 The chemistry of vitrification: why pores disappear—or remain
Most tableware porcelains are based on a triaxial system of clay/kaolin + feldspar + quartz. During firing, the body evolves from a porous compact into a partially glassy, partially crystalline composite. The key transformations include:
- Kaolinite dehydroxylation to metakaolin:
Al2Si2O5(OH)4 → Al2Si2O7 + 2H2O ↑
- Further high-temperature reorganization toward mullite plus silica-rich phases (simplified representation):
3Al2Si2O7 → 3Al2O3 · 2SiO2 + 4SiO2
Here, mullite ((3Al_2O_3 \cdot 2SiO_2)) is the key reinforcing crystal phase in porcelain, while feldspar contributes the liquid/glassy phase that fills voids and promotes densification. Source
The modern materials literature describes porcelain vitrification as a coupled process: feldspar begins melting in roughly the 1000–1100°C interval, liquid phase formation accelerates densification, and mullite crystallization between roughly 1100–1180°C builds an interlocked skeleton that improves dimensional stability. For tableware porcelain, the cited firing route is commonly double firing, with biscuit at 900–1000°C and glost/gloss firing at 1250–1400°C. Source
3.3 Why low absorption improves service performance
A body with low water absorption usually has:
- higher vitrification
- lower connected porosity
- better glaze support
- lower risk of moisture-assisted expansion
- improved resistance to staining and detergent ingress
- more stable long-term appearance and hygiene performance
This is why the <0.5% threshold has become strategically important in premium porcelain and why it appears in both vitrified-product discussion and formal porcelain tableware specification. Source Source
4. Factory Control: How Manufacturers Actually Control Water Absorption
Water absorption is not controlled by one step alone. It is the final outcome of raw material chemistry, particle size distribution, forming density, firing curve, and glaze-body interaction.
4.1 Body formulation control
At the recipe level, the manufacturer must balance three roles:
- Clays / kaolin: plasticity and alumina source
- Feldspar / nepheline syenite / mixed alkali fluxes: liquid phase generation
- Quartz / silica: skeletal stability and shrinkage control
If flux is too low, the body remains under-vitrified and absorption stays high. If flux is too high, the body may overfire, deform, or bloat. If quartz is poorly controlled, thermal expansion mismatch and residual stress can worsen glaze defects even when nominal absorption looks acceptable.
4.2 Milling and particle size control
A coarse or poorly dispersed body does not sinter uniformly. In practice, absorption stability depends strongly on slurry homogeneity, deflocculation consistency, and fineness after milling. The cited comparative porcelain study milled raw batches to a mean particle diameter of roughly 2.7–3.5 μm, illustrating how fine grinding supports densification and predictable firing response. Source
In factory terms, the goal is not “as fine as possible,” but as uniform as necessary. Overly coarse particles leave refractory islands and residual porosity; excessive fines may create forming and drying problems. High-level factories therefore monitor residue, particle-size distribution, slurry viscosity, and specific gravity rather than relying on one single sieve number.
4.3 Forming density and drying uniformity
Whether the item is made by roller-heading, jiggering, isostatic pressing, pressure casting, or slip casting, the green body must be dense and uniform. Local density variation becomes local firing variation. That means one plate rim may mature differently from its center, or one mug handle zone may remain more porous than the cup wall. Uneven drying compounds the problem by introducing microcracks that later act as easy channels for water ingress.
4.4 Firing curve: the decisive control point
The firing curve determines whether the pore network is truly closed down. For tableware porcelain, the published industrial route is commonly:
- Biscuit firing: 900–1000°C
- Glost / gloss firing: 1250–1400°C Source
Inside that window, the real control variables are:
- heating rate through quartz and gas-release zones
- peak temperature
- soak time at maturity
- cooling rate through the glaze-body stress window
A good factory does not simply chase a lower absorption number by raising peak temperature. It seeks the maturity window where absorption is low without pyroplastic deformation, glaze boiling, or excessive residual stress. In strong suppliers, this is managed by SPC charts, kiln profile verification, and lot-by-lot correlation between water absorption, shrinkage, whiteness, and defect rate.
4.5 Testing discipline: internal QC vs external certification
Under ASTM C373, representative specimens are dried to constant mass, commonly at 150 ± 5°C for at least 24 hours, then boiled and soaked before reweighing. The standard also requires at least five representative specimens and defines the weighings for dry, suspended, and saturated conditions. Source
This matters commercially because many factories run quick internal screens that are useful for process control but not equivalent to certified third-party reports. Serious buyers should therefore verify not only the value itself, but also the test method, sample count, conditioning procedure, and whether decoration-fired wares were tested in their final commercial state.
5. Defect Analysis: What Goes Wrong When Water Absorption Is Poorly Controlled
5.1 Underfiring and residual porosity
If firing maturity is insufficient, the body retains too much connected porosity. The immediate symptom is high water absorption; the downstream symptoms are more expensive:
- reduced edge strength
- greater stain pickup at the foot ring
- moisture-assisted discoloration
- lower resistance to dishwasher attack
- higher risk of delayed crack growth
For hotelware and high-turnover tableware, this is a hidden durability cost because failures often appear after repeated service, not during incoming inspection.
5.2 Crazing
Crazing is usually a glaze-body thermal expansion mismatch problem, but water absorption matters because a more porous body is more vulnerable to moisture expansion over time. That post-production expansion can place the glaze into tension and trigger delayed crazing, especially when the body was marginally matured to begin with.
5.3 Pinholes and blisters
Pinholes are not caused by water absorption alone. However, a persistently high absorption value often reveals the same root issue that causes pinholes: an immature firing state with incomplete densification, residual gas release, or poorly managed burnout. In practice, a factory struggling to reach target absorption often also struggles with pinholes, blisters, dry glaze, orange peel, and uneven gloss because the thermal window is not fully under control.
5.4 Dunting, chipping, and glaze support failure
A porous body is mechanically weaker and less supportive beneath the glaze. Repeated thermal cycling or utensil impact can then cause rim chipping, micro-spalling, or crack initiation at local stress concentrators. The danger is especially high in wide-rim plates, stackable bowls, and thin-wall cups where geometry amplifies residual stress.
6. Buyer Decision Framework: Why B2B Purchasers Must Care
For an OEM, distributor, hospitality brand, or private-label importer, water absorption is one of the fastest ways to judge whether a supplier understands ceramic body engineering or merely reaches an acceptable appearance on the shipment date.
6.1 What a smart buyer should ask for
A serious buyer should request:
- a recent water absorption report with method clearly stated
- the standard used: e.g., ASTM C373 or the method referenced by ISO 5644
- whether the result is from body test pieces or finished commercial ware
- supporting data on thermal shock, dishwasher durability, lead/cadmium release, and defect rate
Low absorption alone does not guarantee food safety. A dense porcelain body can still fail if glaze chemistry is poor. That is why absorption must be evaluated together with food-contact compliance, not instead of it. Source
6.2 How to interpret supplier capability
As a practical B2B rule:
- A supplier who only says “our porcelain is low absorption” is making a sales claim.
- A supplier who provides test method, sample size, production lot, firing date, and trend stability is showing process control.
- A supplier who can explain the link between body recipe, kiln curve, shrinkage, and defect map is usually operating at a much higher technical level.
In custom tableware projects, that difference matters because decoration, shape complexity, wall thickness, and glaze color can all shift the maturity window. High-level factories understand that water absorption is not a static catalog value; it is a process capability indicator.
6.3 Procurement red flags
Buyers should be cautious when:
- absorption reports lack a standard reference
- only one specimen was tested
- the supplier reports body data but avoids final-ware data
- the supplier cannot explain crazing history after dishwasher or microwave use
- the supplier treats porcelain, stoneware, and ceramic as interchangeable marketing words
ISO 5644’s porcelain criterion of <0.5% water absorption is especially useful here because it gives buyers a specification anchor when discussing premium porcelain tableware. Source
7. International Standards and Compliance Benchmarks
7.1 ASTM C373
This is one of the most widely recognized methods for determining water absorption, bulk density, apparent porosity, and apparent specific gravity of fired ceramic whitewares and related products. Its significance statement is important: these measurements are tools for evaluating the degree of maturation and the structural suitability of a ceramic body. Source
7.2 ISO 5644
This is directly relevant to porcelain tableware. Its official summary states that porcelain tableware shall have water absorption less than 0.5%, determined according to the referenced method. For B2B purchasing, this is one of the clearest specification references for premium porcelain positioning. Source
7.3 ISO 6486-1:2019
This standard covers the test method for release of lead and cadmium from ceramic ware, glass ceramic ware, and glass dinnerware intended for food contact. It does not replace water absorption testing, but it is essential for export compliance because a low-absorption body still must pass food-contact migration requirements. Source
7.4 FDA Lead Guidance for Ceramic Ware
FDA’s compliance policy guide for ceramic foodware gives category-specific lead action levels, including 3.0 μg/mL for flatware, 2.0 μg/mL for small hollowware other than cups/mugs, 1.0 μg/mL for large hollowware other than pitchers, and 0.5 μg/mL for cups/mugs and pitchers. This is a reminder that body maturity and glaze safety are related in quality management, but they are not the same test. Source
7.5 FDA Cadmium Guidance for Ceramic Ware
FDA also maintains a dedicated compliance policy guide for cadmium contamination in ceramic ware. For exporters serving the U.S. market, this should be checked alongside lead migration, especially for decorated mugs, reactive glazes, and dark-colored food-contact surfaces. Source
8. Final Insight
In ceramic tableware, water absorption is not just a number—it is a condensed fingerprint of body maturity, pore structure, kiln control, and long-term durability. If the value is too high, the body is telling you that its microstructure is still open, its vitrification is incomplete, and its lifetime reliability is compromised. If the value is low but unstable from lot to lot, the factory has not yet mastered repeatability. The best suppliers are not those with the lowest single test result, but those who can hold a controlled low value consistently, while also passing glaze fit, thermal shock, and food-contact compliance.
For B2B buyers, that is the real takeaway: water absorption is one of the fastest technical shortcuts to separating appearance-based suppliers from process-capable manufacturers.
References
- ASTM C373 official summary – Standard test methods for water absorption and associated properties
https://www.astm.org/c0373-18.html - Accessible ASTM C373 text extract / PDF copy – formula, weighing sequence, drying and boiling conditions
https://downloads.regulations.gov/EPA-R09-OAR-2016-0711-0003/attachment_11.pdf - ISO 5644 official summary – porcelain tableware specification, including <0.5% water absorption
https://www.iso.org/obp/ui/en
If you have any questions or need to custom dinnerware, please contact our Email:info@gcporcelain.com for the most thoughtful support!









